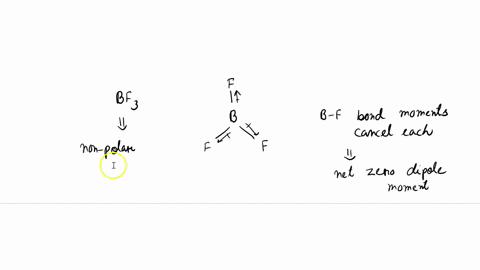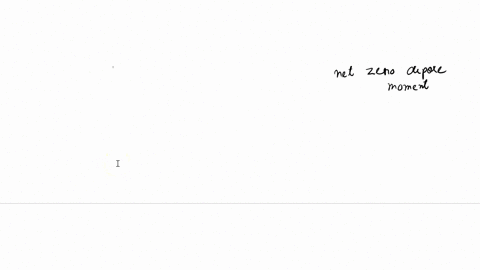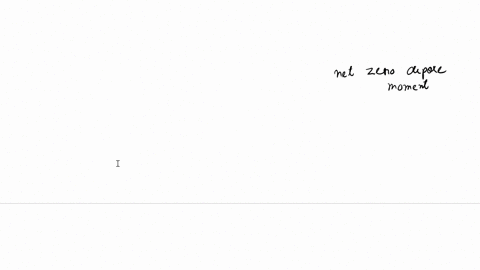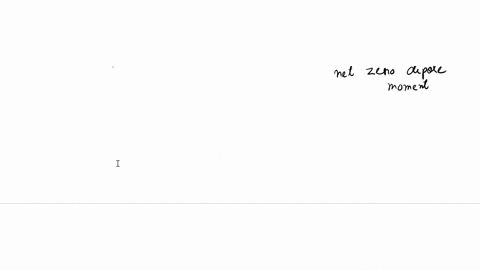
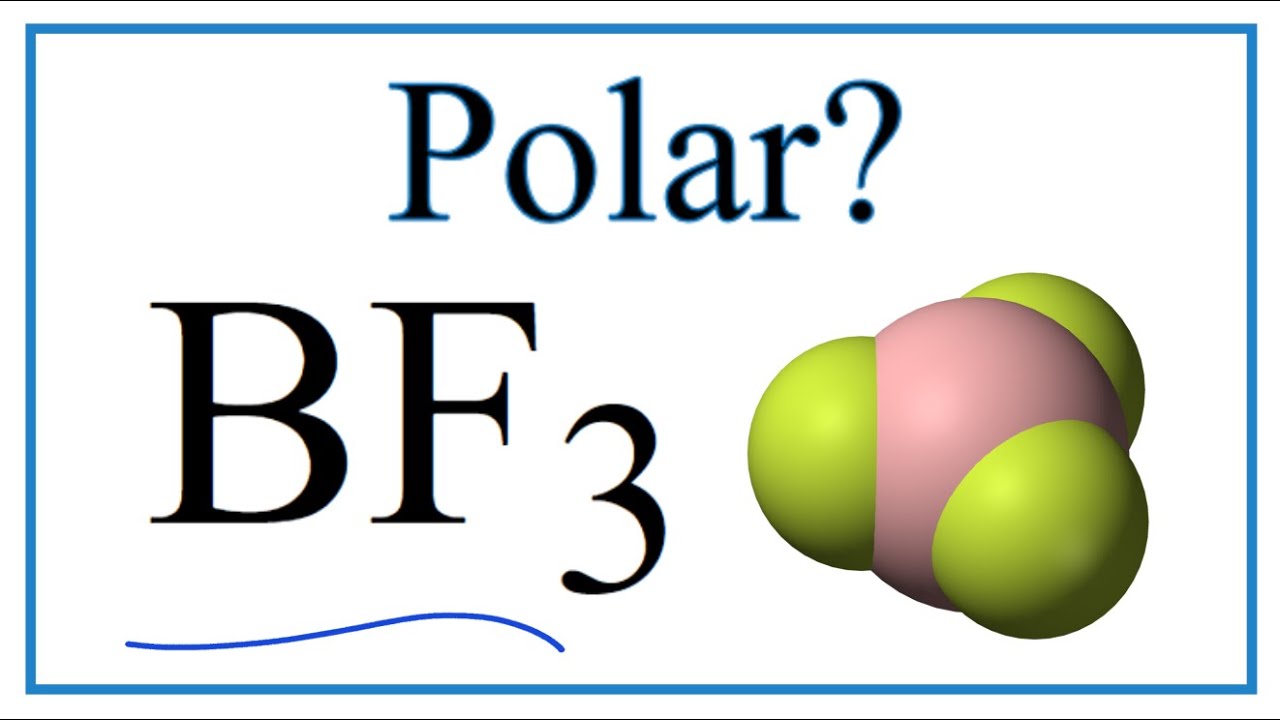
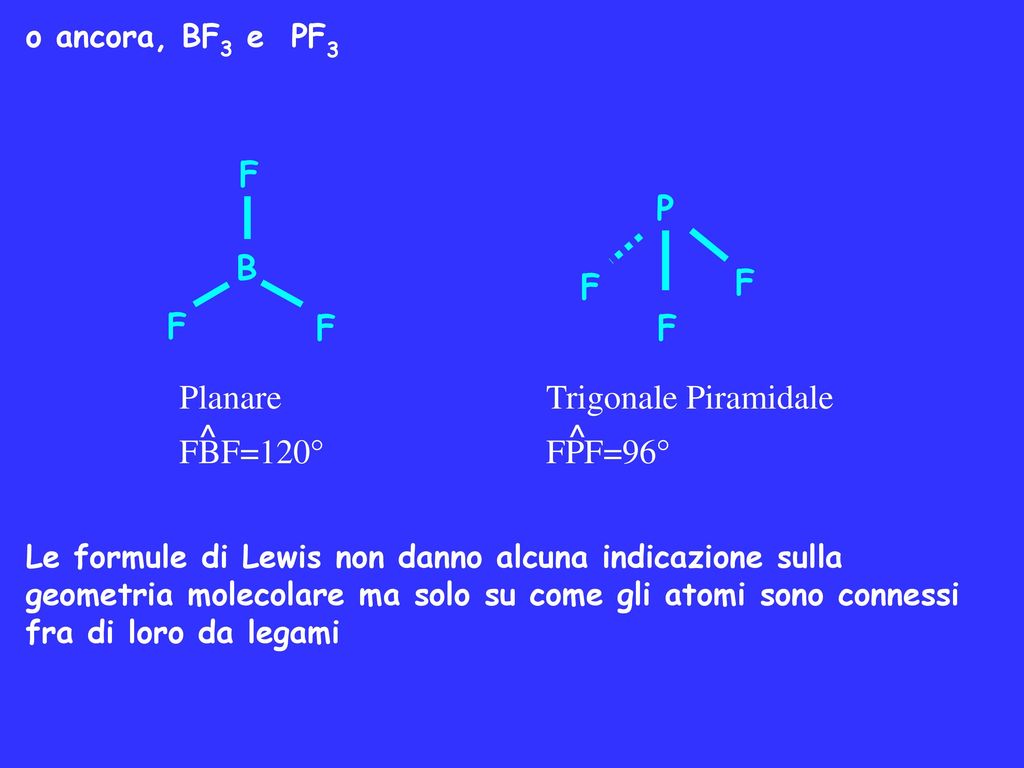
SOLVED: (a) Is the molecule BF3 polar or nonpolar? (b) If you react BF3 to make the ion BF3^2-, is this ion planar? (c) Does the molecule BF2Cl have a dipole moment?
✓ Solved: The molecules BF3, CF4, CO2, PF5, and SF6 are all nonpolar, even though they contain polar...
Both BF3 and NF3 are covalent compounds but NF3 is a polar compound while BF3 is non-polar. How can you explain it? - Quora










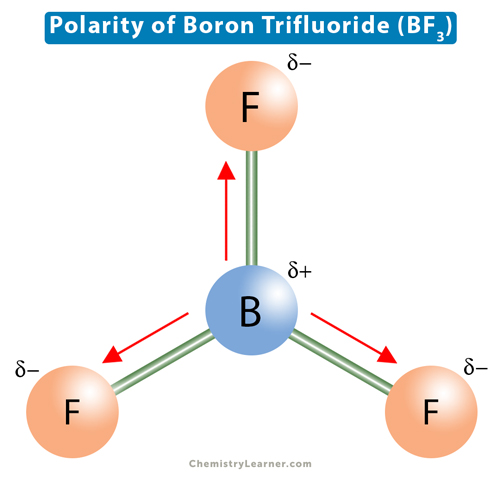
![Best Overview on: BF3 Polar or Nonpolar [#1] - Science Education and Tutorials Best Overview on: BF3 Polar or Nonpolar [#1] - Science Education and Tutorials](http://sciedutut.com/wp-content/uploads/2021/05/bf3-1.png)