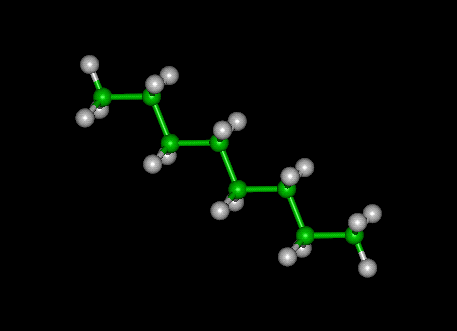
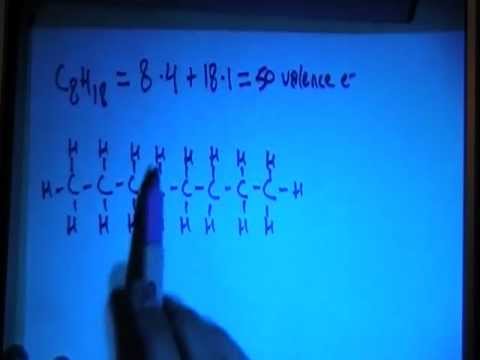Molecular structure of type II kerogen fragment (left), nonpolar oil... | Download Scientific Diagram

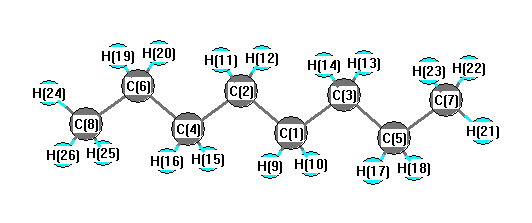
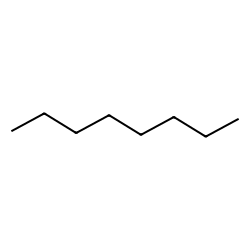
a) Are there polar bonds in Hexane? (Show EN for each of the bonds in the molecule) b) What is the geometric shape of Hexane (VSEPR)? c) Is this molecule polar or
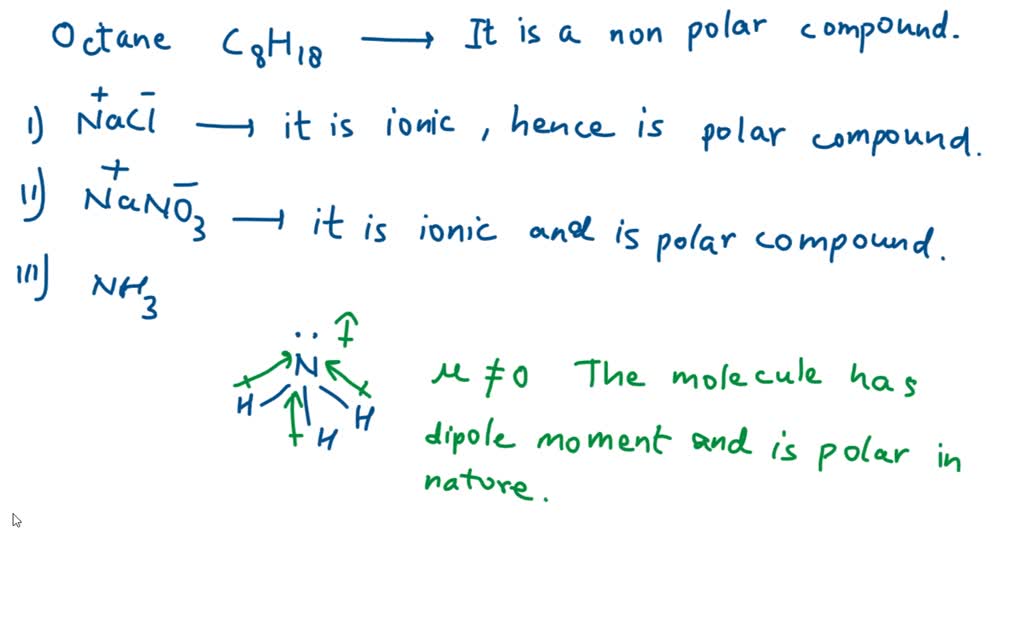
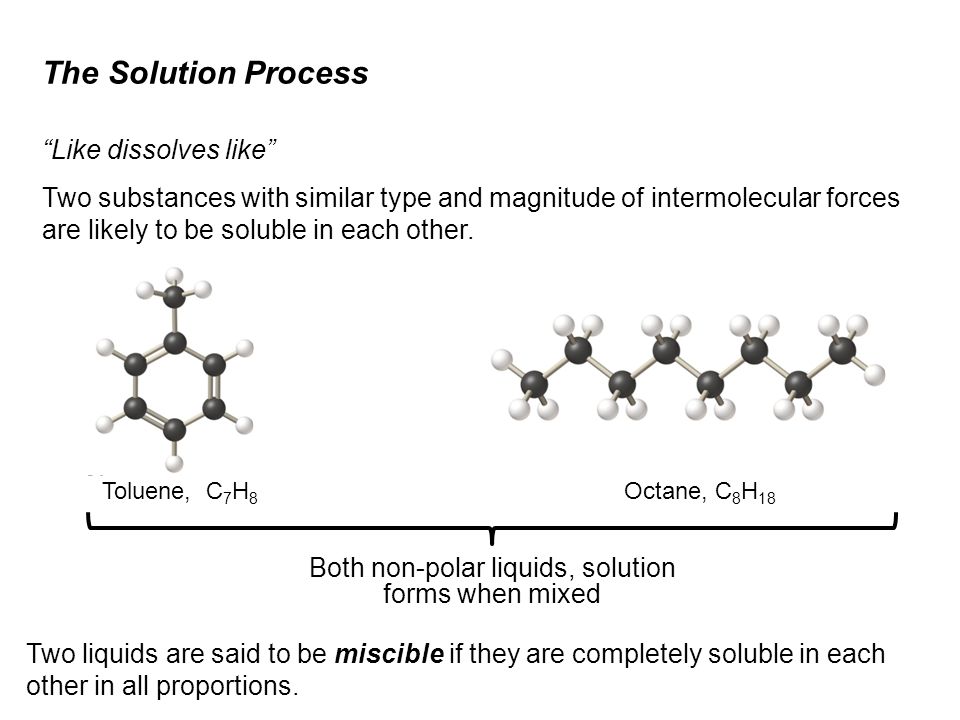
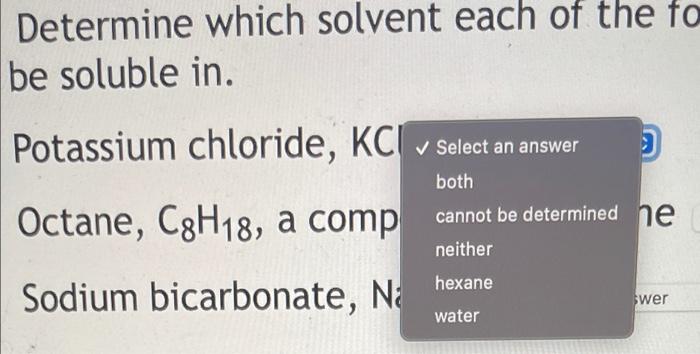
SOLVED: 37. Which of the following should be most soluble in octane (C8H18)? NaCl, NaNO3, NH3, CH3OH, Br2.
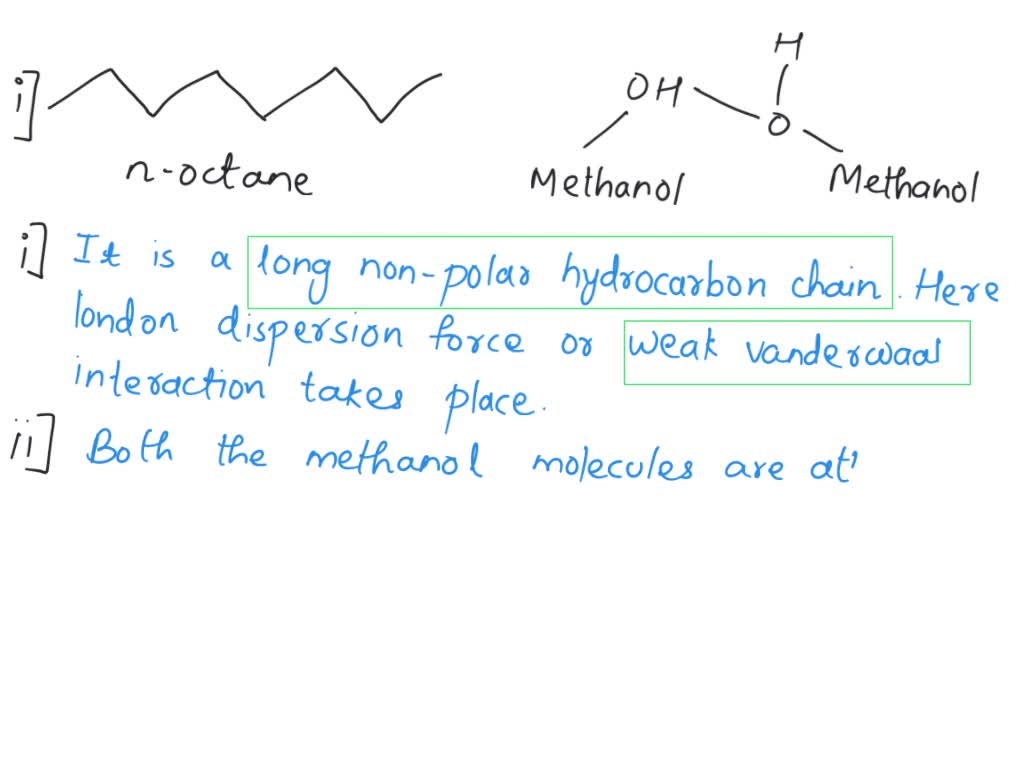
SOLVED: For n-octane (C8H18) and methanol (CH3OH), determine the predominant type of intermolecular attractions for the pure liquids (dispersion, permanent dipole, or hydrogen bonding). Predict whether these intermolecular attractions will be strong

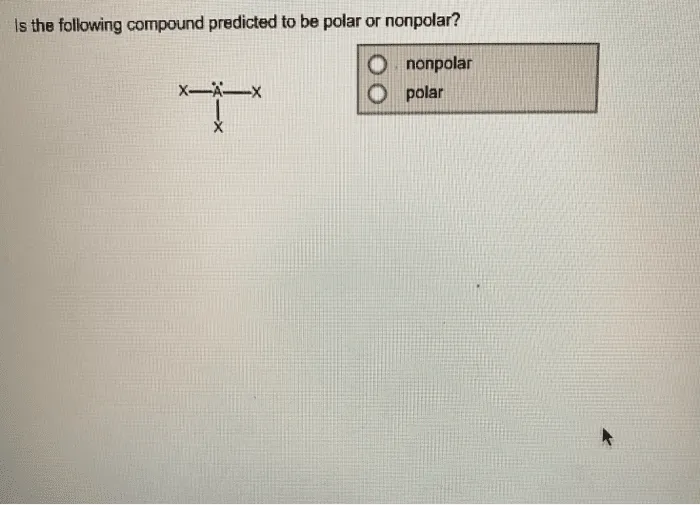
PO43- Lewis structure, molecular geometry or shape, bond angle, hybridization, polar vs non-polar | Molecular geometry, Molecular, Vsepr theory