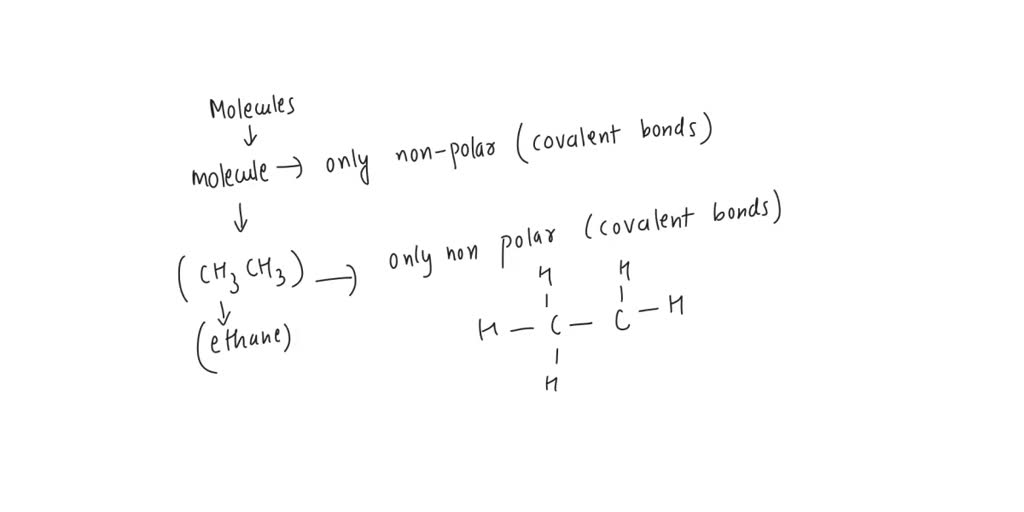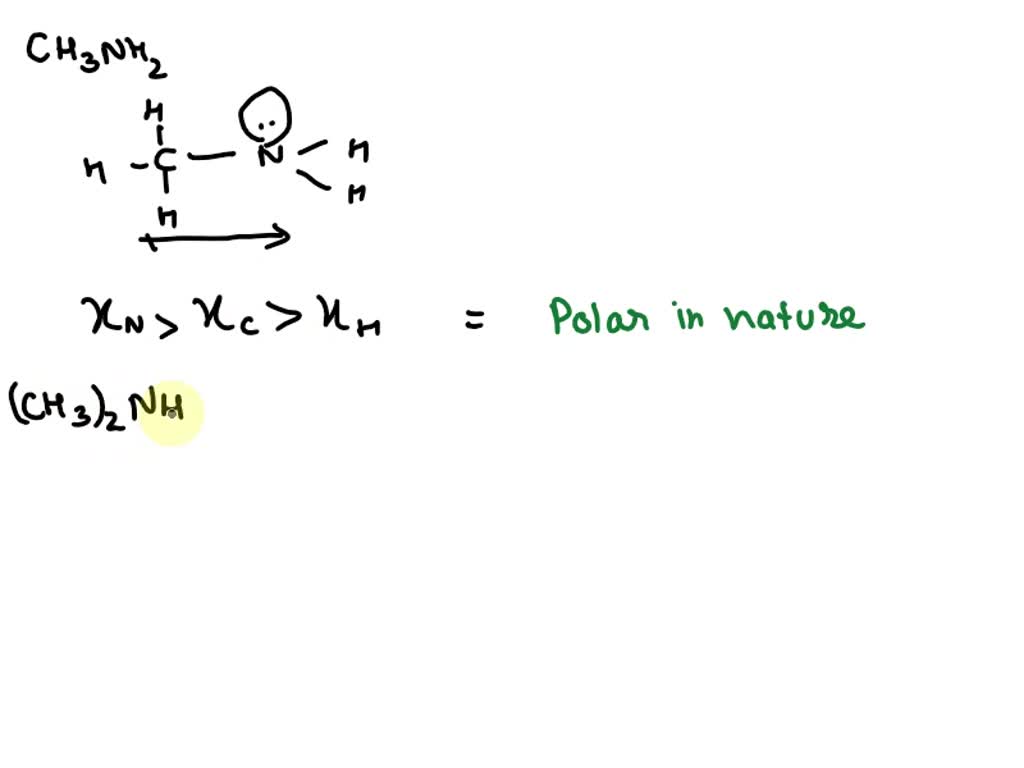CH3NH2 Lewis Structure, Molecular Geometry, Hybridization, Polar or Nonpolar - CH3NH2 Lewis - Studocu

Is CH3NH2 polar or non-polar? - Polarity of CH3NH2 | Molecular geometry, Covalent bonding, Electron configuration

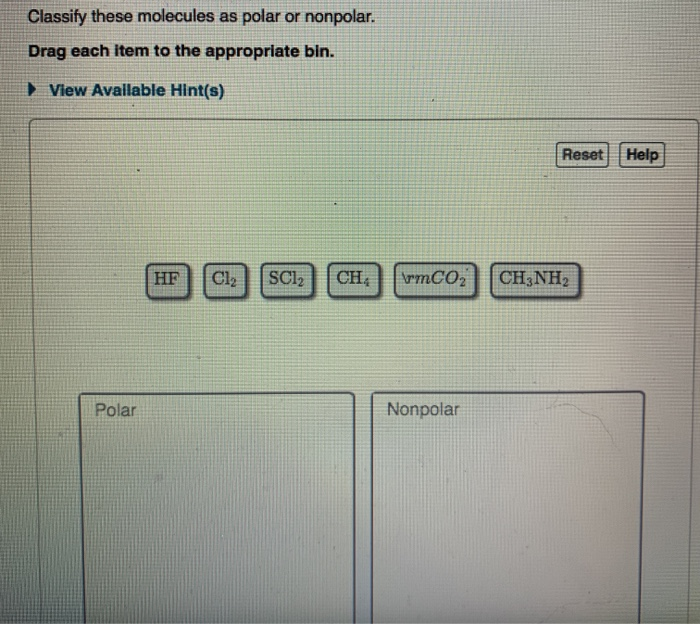
SOLVED: Text: Identify the polarity of these molecules and classify them as polar or nonpolar. Drag each item to the appropriate bin. View Available Hint(s) HBc BBeg CBr4 CH3NH2 H2O Polar Nonpolar

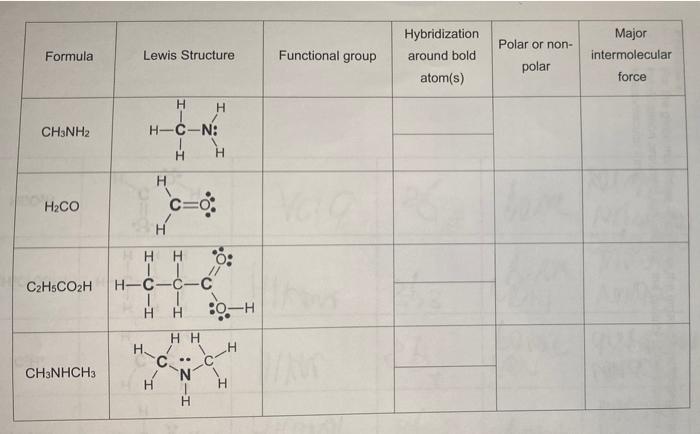
SOLVED: 25. Indicate whether each of the following compounds are polar or non-polar. a) CHCl2 - polar b) CF4 - non-polar c) H2CNH - polar d) CH3OH - polar 26. Indicate whether

CH3NH2 Lewis Structure, Molecular Geometry, Hybridization, Polar or Nonpolar - CH3NH2 Lewis - Studocu

CH3NH2 Lewis structure, molecular geometry, hybridization, bond angle | Molecular geometry, Molecular, Molecular shapes
![SOLVED: 3. For the molecule of methylamine, CH3NH2, do the following: a) Draw the Lewis structure. Show your work. [A-2] b) Use the VSEPR theory to predict the shape around the two SOLVED: 3. For the molecule of methylamine, CH3NH2, do the following: a) Draw the Lewis structure. Show your work. [A-2] b) Use the VSEPR theory to predict the shape around the two](https://cdn.numerade.com/ask_previews/9cee942f-600a-46e5-8ea2-aa1989ba2d4e_large.jpg)
SOLVED: 3. For the molecule of methylamine, CH3NH2, do the following: a) Draw the Lewis structure. Show your work. [A-2] b) Use the VSEPR theory to predict the shape around the two
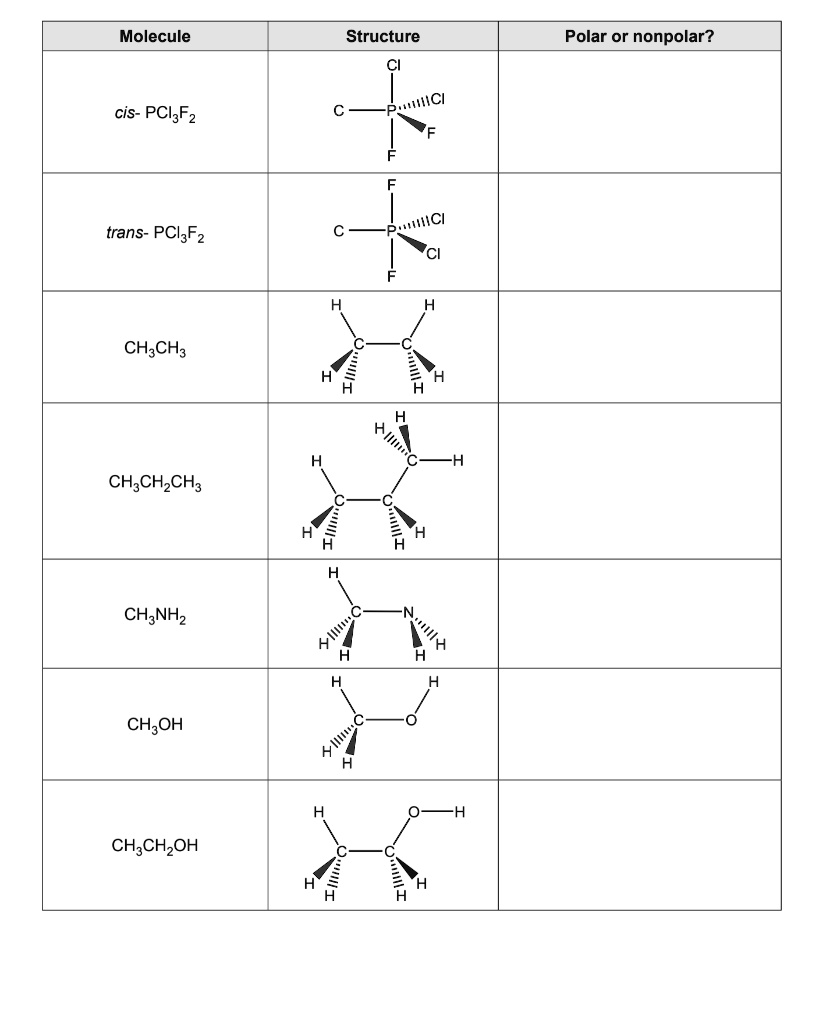
SOLVED: Molecule Structure: Polar or Nonpolar? cis-PCIF3 trans-PCIF2 CH2CH2 CH2CH2CH3 CH2NH2 CH2OH CH2CH2OH PaCl ICl 01 OS 1 0