CO32- Lewis structure, molecular geometry, bond angle, formal charge, hybridization | Molecular geometry, Molecular, Electron configuration

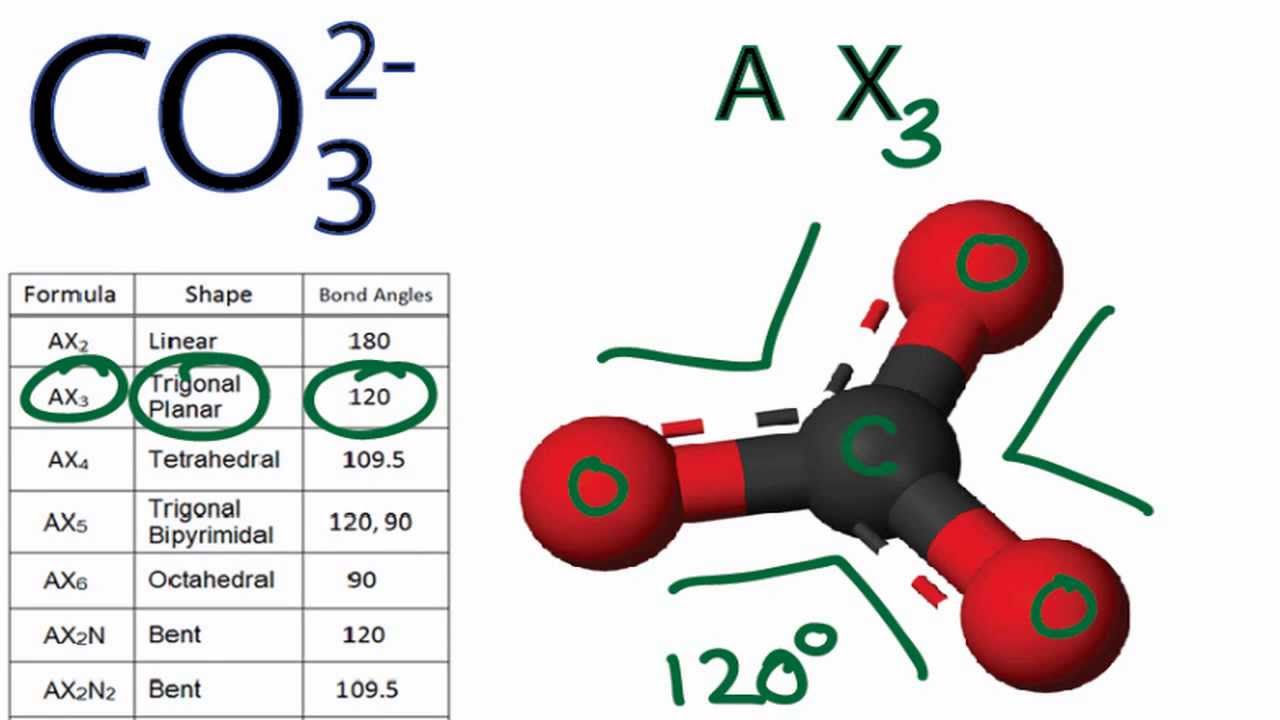
Is CO3 2- Polar or Nonpolar (Carbonate ion) | Is CO3 2- Polar or Nonpolar (Carbonate ion) Carbonate ion is a polyatomic ion with the formula CO3-2. It is a carbon oxoanion.

Amongst NO_{3}^{-},AsO_{3}^{3-},CO_{3}^{2-},ClO_{3}^{-},SO_{3}^{2-} and BO_{3}^{3-} the non-polar species are:SO_{3}^{2-}, NO_{3}^{-} and BO_{3}^{3-}AsO_{3}^{3-}, CIO_{3}^{-} and SO_{3}^{2-} CO_{3}^{2-},SO_{3}^{2-} and BO_{3}^{3-}NO_{3}^{-}, CO_{3}^{2 ...

Is CO3 2- Polar or Nonpolar (Carbonate ion) | Is CO3 2- Polar or Nonpolar (Carbonate ion) Carbonate ion is a polyatomic ion with the formula CO3-2. It is a carbon oxoanion.
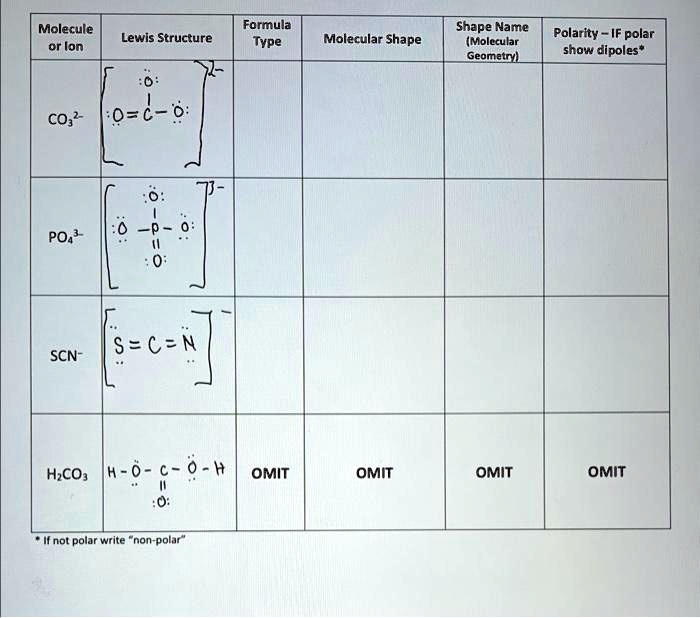
SOLVED: Molecule Formula Type Molecular Shape Shape Name (Molecular Geometry) Lewis Structure Polarity - IF polar show dipoles* 0 CO2 O=C=O 0=d- 0=d- 1 0 PO43- N≡S SCN- HCO2- H-O-H 1 1
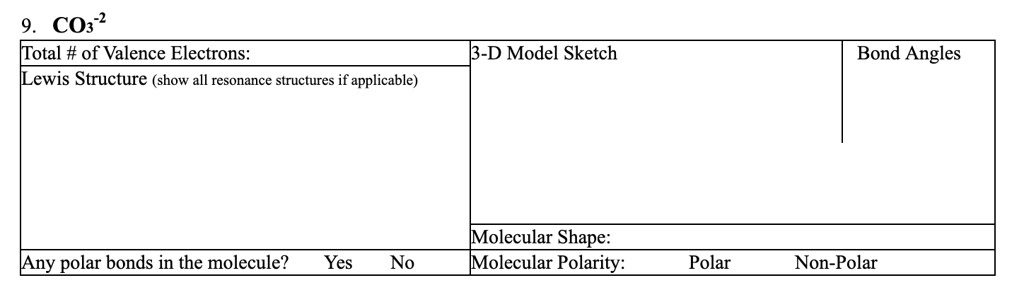
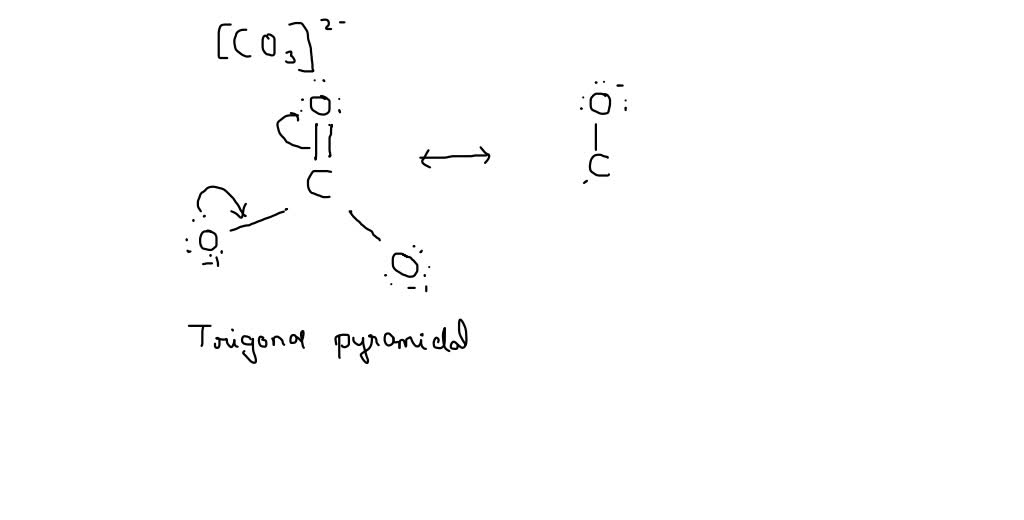
Give the correct Lewis structure (octet theory included) for CO32-. From this structure, give the number of bonded pairs and lone pairs. | Homework.Study.com